Atmospheric humid air enters a coolng tower at a volumetrix

The cooled water moves back to the initial source of heat generation and, further, the cycle repeats.
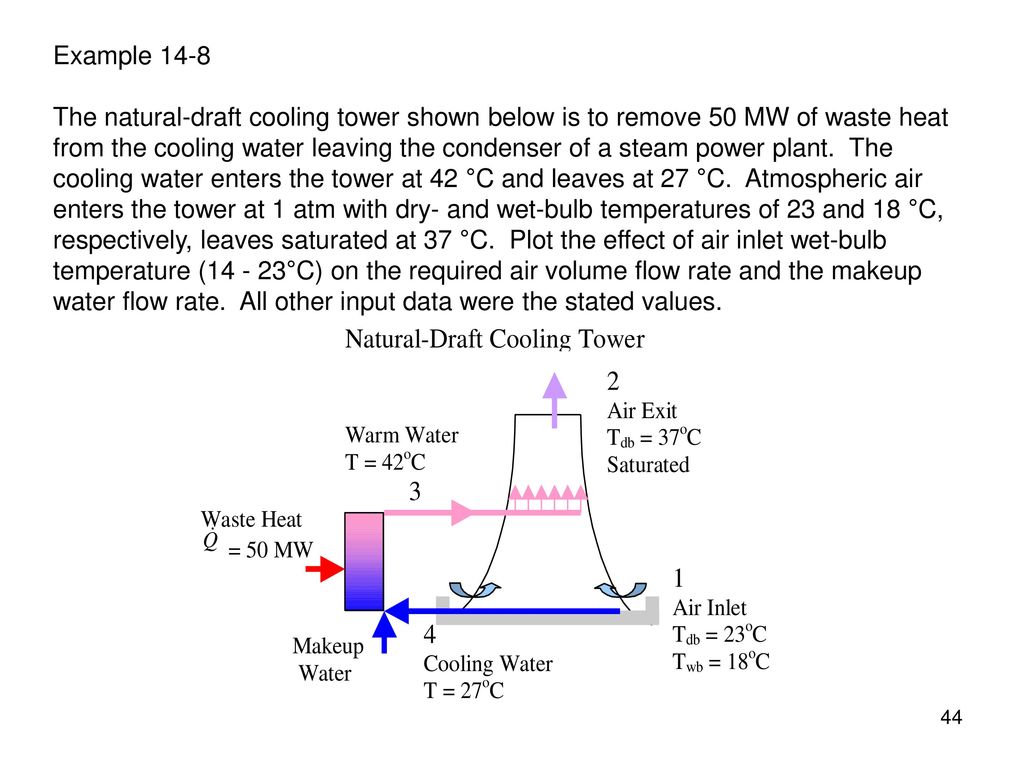
Water loses its heat as the evaporation proceeds and this warm air then enters the tower sump present at the bottom and the warm air is let out. (b) Make Up Water = ma (W2 − W1 ) = ( 3.244 )( 0.0766 − 0.0208) = 0.Fig:1.Schematic Diagram of a Cooling Tower System Image Courtesy: AranerĪ large volume of air is constantly flowing through the large fans present in the tower system. M3 − m4 = ma (W2 − W1 ) m4 = m3 − ma (W2 − W1 )īy energy valance a h1 + m 3h3 = m a h2 + m 4 h4 m Determine (a) the mass flow rate of air entering, and (b) the quantity of make-up water required. Air enters the tower at 32 C dry bulb and 27 C wet bulb temperatures and leaves at 50 C and 90% relative humidity. A cooling tower receives 6 kg/s of water of 60 C. (1) mah1 + m3 h3 = mah2 + m4 h4 By mass balance Volume of air necessary to cool 1 kg of water = V1 = mav1 To solve for ma when m3 =1 kg By energy balance: Eq. It is desired to determine (a) the volume and mass of air necessary to cool 1 kg of water, and (b) the quantity of water that can be cooled with 142 cu m per minute of atmospheric air. Air at 29 C and 47% RH also enters the cooling tower and leaves at 46 C fully saturated with moisture. In a cooling tower water enters at 52 C and leaves at 27 C. H4 =129.70 kJ kg ∴ t4 = 31.9 C - exit water temperature. To solve for h4 : ah1 + m 3 h3 = m 4 h4 + m ah2 m

4: To solve for m By Mass Balance: 3 +m a + W1m a =m 4 +m a + W2 m a m Tdb2 = 36 C saturated V1 =115 m3 s tdb1 = 32 C
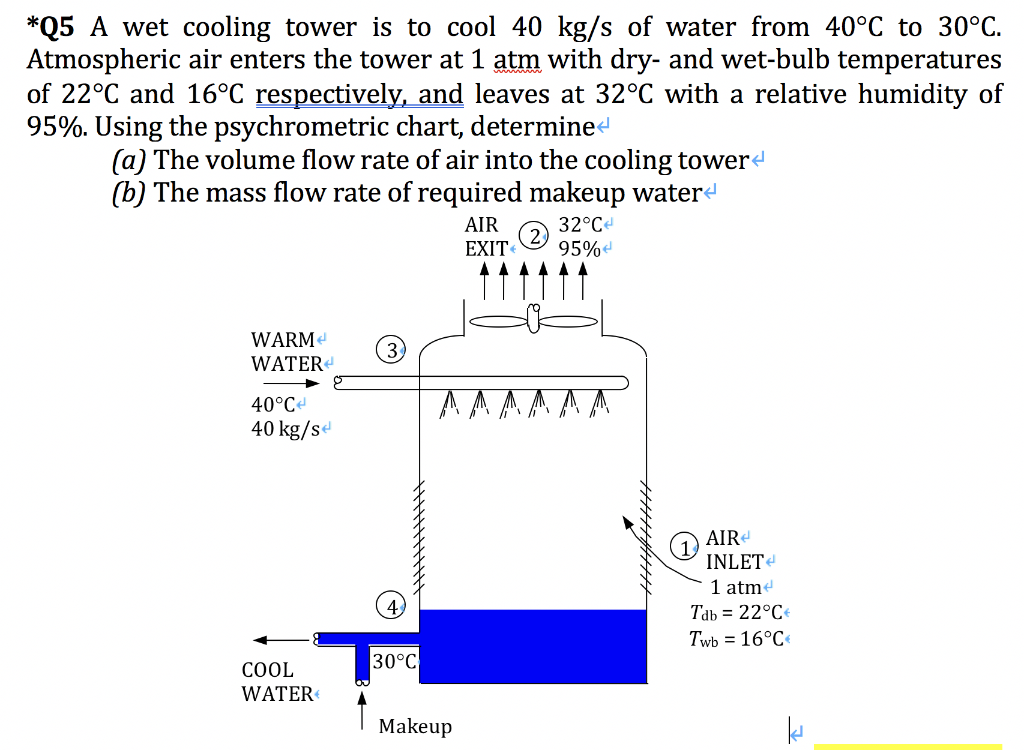
If the tower receives 200 kg/s of water at 40 C, what will be the exit temperature of the cooled water? Solution: A mechanical-draft cooling tower receives 115 m3 per second of atmospheric air at 103 kPa, 32 C dry bulb temperature, 55% RH and discharges the air saturated at 36 C.
